Photos / Sounds
What
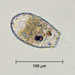
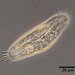
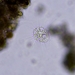
Caenomorpha medusulaObserver
peptolabDescription
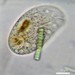
Caenomorpha medusula PERTY, 1852 from the superficial river edge benthos of the freshwater segment of the estuarine Peconic River. Imaged in Nomarsdki DIC on Olympus BH2S using Splanapo 40 0.95 and Splan 100 1.25 oil objectives plus variable phone camera cropping on Samsung Galaxy S9+.
This metopid ciliate is a fast swimmer and often difficult to track, but also holds on to the substrate thigmotactically with the dorsally located long cirri. It is an inhabitant of saprobic anoxic decaying benthic organic debris. My population averages 100 um in total length including the single long caudal spine. There are three macronuclear nodules with two sometimes incompletely separated and a single micronucleus. There is an anterior refractile granular agggregate. The cytoplasm contains diverse rod-shaped bacterial symbionts. The contractile vacuole was rather large in diastole in some individuals. All of these features are in agreement with the most recent redescription by Li et al 2017 (1).
Li et al 2017 provide the most recent and detailed redescription of the species. In describing the most important characters, they write: "Although this species has been studied for more than 150 years, species identification remains problematic because many descriptions are based only on live materials without redescription of the ciliature. Furthermore, some features, e.g. body shape, appear to vary among individuals in different environments, but, nonetheless, have traditionally been used as key characters in the taxonomy of Caenomorpha. The original description of Caenomorpha medusula by Perty (1852) was rather superficial and failed to note some important features (e.g. the numbers of adoral membranelles, bell kineties and spines, etc.), which renders the identification of this organism difficult. However, according to the original and subsequent investigations, this species can be recognized by a combination of the following characters: (i) multiple macronuclear nodules; (ii) two unequal-length bell kineties; (iii) one conspicuous posterior spine" (1).
"Description: Body medusoid, covered with a transparent rigid pellicle, 112–125 × 50–65 um in vivo with a ratio of length to width about 2:1. Posterior spine slender, about 45–55 um long in vivo; ratio of spine length to body length about 0.4. Plump rod-shaped epibiotic bacteria often found in US population but not found in China population. Cytoplasm clear and colourless, with some dark globules (1–2 m across) and an aggregate of transparent granules in anterior body part, rod-like bacteria exist in cytoplasm in China population, very likely endosymbionts. Edges of preoral bell never adjoin closely to posterior body; peristome narrow, deep funnel-shaped. Cytostome near base of spine; undulating membrane recognizable after staining, length of membrane about 50 m long. Contractile vacuole located near base of spine, about 15–20 m in diameter, pulsates at intervals of 3–5 min. Three (41 of 60 cells [Chinese population], 36 of 45 cells [US population]), four (19 of 60 cells [Chinese population], 8 of 45 cells [US population]) or five (1 of 45 cells [US population]) macronuclear nodules, usually ovoid or ellipsoidal, arranged in line, located in center of cell, sometimes incompletely separated; one micronucleus, ellipsoidal, near macronuclear nodules. Movement leisurely, spiraling while rotating around the long axis of the body" (1).
"Two strongly thigmotactic bell kineties about 68 um and 35 um long, respectively, located in anterior part of dorsal side of cell, consist of about 94 and 56 cirri (Chinese population), and 108 and 59 cirri (US population) respectively (n = 21); cirri in each kinety arranged in indistinct zig-zag pattern. Perizonal stripe beginning near anterior end of cell, about 6.4 um wide at middle part, composed of 114–180 kineties (114–169 in China population, 123–180 in US population), spiraling 450 degrees around axis; each kinety inclined about 60 degrees to edge of shield; longest kinety (at middle of stripe) composed of about 15 pairs of kinetosomes in both populations, whereas ones near oral region with only two pairs of kinetosomes. Adoral zone composed of 41-67 membranelles (Chinese population), each with three or four rows of kinetosomes, spiraling 360 degrees around body axis from near the distal perizonal stripe, terminates near cytostome. Undulating membrane on undersurface of preoral bell (i.e. roof of peristomial region), about 50 m long. Cilia on base of spine, invariably arranged in two short kineties, each 10–15 m long, composed of about 25 kinetosomes each. The two spine kineties inclined about 20 degrees to each other, converge posteriorly" (1).
Jankowski provided an older but elegant description: "Body medusoid, covered with a clear transparent rigid pellicle that looks like an armour; body length, without a spine, is 70-90 x 65-70 ul, spine 30-35 um long. The shield bears no ciliary meridians except for those of a perizonal ciliary stripe. Instead. it bears two groups of long thick flexible cirri, with 8-10 cirri in each group: they are perfectly seen from the left side. These cirri are highly thigmotactic- one can frequently observe a prolonged adhesion of the animals to sapropelic particles by the aid of these cirri, while both perizonal cilia and adoral membranelles continue their activity. The edges of the shield never adjoin closely to the body surface; instead, a narrow deep funnel may be observed between them. The perizonal ciliary stripe occupy the margin of the shield; it is composed of 5 ciliary rows not separated into two groups, unlike that of Metopidae (where it includes 2 upper and 3 lower kinetics). The PCS of Caenomorpha looks like a wide densely ciliated field composed by a number of short oblique ciliary rows with 5 kinetosomes each. It serves for both feeding and movement in this genus. The synchronous beating of the perizonal cilia and adoral membranelles produces a rotation of the swimming animal and, in addition, creates the intense water current along the buccal groove. driving the food-particles into the intrastomium. The cytostome in C. medusula occupies a typical for all the caenomorphids posterior position with a thin tubular cytopharynx raising right up into the anterior body part, where colourless food vacuoles are concentrated" (2).
- Description of two species of caenomorphid ciliates (Ciliophora, Armophorea): Morphology and molecular phylogeny. Song Li, William A. Bourland, Saleh A. Al-Farraj, Lifang Li and Xiaozhong Hu. European Journal of Protistology 61 (2017) 29–40.
- Morphology and Evolution of Ciliophora. III. Diagnoses and Phylogenesis of 53 Sapropelebionts, Mainly of the order Heterotrichida. ANATOL W. JANKOWSKI. Arch. Protistenk. Bd. 107, S. 185-294 (1964)
Observer
tomas_cedhagenDescription
Distance between crossing lines = 5 mm.
They were picked on the shore.
Photos / Sounds
What
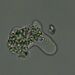
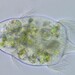
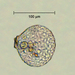
Stentor pyriformisObserver
peptolabDescription
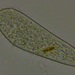
Stentor pyriformis Johnson 1893 from the superficial benthos of the river edge of the freshwater segment of estuarine river Peconic River. Imaged in Nomarski DIC on Olympus BH2S using SPlan 10 NA 0.30, SPlanapo 20 NA 0.70, SPlanapo 40 NA 0.95 and SPlan 100 NA1.25 oil objectives plus variable phone camera cropping on Samsung Galaxy S9+. The cells were visible with the naked eye accumulating on the surface of the water at the side of the sample container. Cells measure from 650-800 um when fully extended. The cytoplasm is densely filled with green algal zoochlorella symbionts. There are also much smaller colorless cortical and cytoplasmic granules. The dense population of zoochorellae obscured the internal morphology of the stentors. Macronuclei could only be observed in markedly squashed cells by evaporation of water from under the coverglass. Most cells had two spherical macronuclei, occasionally three. The morphology of my population was identical to that described by Hoshina et al 2021, however their population was smaller than mine at 220-500 um and their macronuclear count also differed from my population which showed 2 MA: "The average number of macronuclei was 6.1 (range 4–10, n = 9) for freshly obtained samples, whereas four-year cultured cells contained only one or two" (1).
"S. pyriformis is a poorly described species, although S. pyriformis is clearly distinguishable from other Stentor species. The species was first described in 1893 and then appeared in a microbiota report in 1908. However, its next appearance was not until 1994, in the study on revision of the genus by Foissner and Wolfl. As described in the original literature, difficulties in the cultivation of this species may have hindered the research on this species. In Japan, S. pyriformis can be found only in highland highly oligotrophic moors, suggesting that intracellular symbiotic algae would help this species of Stentor survive in such a harsh environment. S. pyriformis was described by Johnson in 18936. This algae-bearing Stentor has separated spherical macronuclei without pigmentation, which certainly differentiates it from other Stentor species. While the most common algae-bearing Stentor, S. polymorphus assumes a slender trumpet shape (often shortened), S. pyriformis never resembles such a slender trumpet, but assumes a pear or short conical shape, even when it is swimming. Among algae-bearing Stentor spp., S. polymorphus and S. pyriformis only are considered colorless species, whereas colored species are S. amethystinus, S. fuliginosus, S. araucanus, and S. tartari. Therefore, S. pyriformis is a clearly discernible species; however, it remains underexplored" (1).
"Cells of S. pyriformis were broadly trumpet-shaped, usually 220–500 × 120–300 µm. This length–width ratio did not change significantly between the cells attached to something and swimming. The cells were colored green due to their endosymbiotic green algae that were distributed along the whole body. A large number of tiny transparent vesicles were present along the ciliary rows immediately under the cell surface. To see the contents, the crushed cells were observed. Symbiotic algae appeared to be typical Chlorella-like algae, but no dividing alga was observed. The algal cells appeared more vividly green when compared to those in P. bursaria, suggesting that they are richer in photosynthetic pigments. The symbiotic algae in S. pyriformis had the same size and morphology as those in P. bursaria. Macronuclei were, in general, large and spherical (ø 20–35 µm). The average number of macronuclei was 6.1 (range 4–10, n = 9) for freshly obtained samples, whereas four-year cultured cells contained only one or two. Micronuclei could not be identified" (1).
My population had two macronuclei rarely three in contrast to the populations of Hoshina et al which had an average of 6.1 macronuclei (range 4-10). Also, Hoshina et al 2021 (1) did not identify micronuclei in their multimodality study of S. pyriformis which included electron microscopy. However, the study of Walker 1908 (2) describes the presence of two macronuclei with multiple small micronuclei scattered within the macronucleus. I detected similar small nodules within the macronuclei to those depicted by Walker 1908 but I am uncertain if these are nucleoli or micronuclei. Hishina et al 2021 describe " a large number of transparent vesicles were present along the ciliary rows immediately under the cell surface (1) which I also found. In addition, Hoshina et al 2021 describe numerous starch granules in the cytoplasm which I believe are also present in my samples. These granules are more than twice the size of the subcortical vesicles.
- Characterization of a green Stentor with symbiotic algae growing in an extremely oligotrophic environment and storing large amounts of starch granules in its cytoplasm. Ryo Hoshina, Yuuji Tsukii, Terue Harumoto & Toshinobu Suzaki. Scientific Reports | (2021) 11:2865 | https://doi.org/10.1038/s41598-021-82416-9
- Observations on the Micro-Fauna of an Oregon Pond. Elda R. Walker. Transactions of the American Microscopical Society, Vol. 28, Twenty-Ninth Annual Meeting (Sep., 1908), pp. 75-84
Photos / Sounds
Observer
peptolabDescription
Cryptomonas (Chilomonas) paramecium Ehrenberg 1832. A bloom from the acidic freshwater kettle pond Chatfield's Hole fed with boiled wheat seeds. Imaged in Nomarski DIC on Olympus BH2S using SPlan 100 1.25 oil objective plus variable phone cropping on Samsung Galaxy S9+. As Jara Kubin points out, the correct name after molecular analyses is Cryptomonas paramecium, the species is a regular member of the genus, but without chlorophyll.
Hoef-Emden, K. & Melkonian, M. (2003). Revision of the genus Cryptomonas (Cryptophyceae): a combination of molecular phylogeny and morphology provides insights into a long-hidden dimorphism. Protist 154: 371-409
Genus: Similar to Cryptomonas in general body form and structure, but colorless; without pyrenoid; "cytopharynx" (gullet) deep, lower half surrounded by granules (ejectisome/trichocyst); one contractile vacuole anterior (Kudo, 1966). Chilomonas does not have chromatophores (pigment-containing structures) and lives by ingesting organic matter.
Species: 20-40 μm long. Posteriorly narrowed; slightly bent 'dorsally'; 30-40 μm long, 10-15 μm wide; widely distributed in stagnant water (Kudo, 1966).
Free-swimming, biflagellate monads, generally obovoid often with a recurved posterior sometimes produced to acutely pointed; not known to produce palmelloid colonies. A longitudinal furrow extends posteriorly from the vestibulum and transforms into a sack-like gullet lined with many rows of ejectosomes. With a single nucleomorph but lacking a chloroplast and pyrenoid; with a prolific accumulation of peripheral starch granules. Periplast with a laminate inner component and a superficial layer of fine fibrillar material. Sexual reproduction is unknown; reproduction being through simple cell division. Cyst formation is unknown.
Photos / Sounds
What
Stentor igneusObserver
peptolabDescription
Stentor igneus Ehrenberg, 1838 from the acidic freshwater kettle pond Chatfield's Hole after feeding the culture with boiled wheat seeds. Imaged in Nomarski DIC on Olympus BH2S using SPlanapo 40 0.95 and SPlan 100 1.25 oil objectives plus variable phone cropping on Samsung Galaxy S9+.
Rose-colored or colorless; macronucleus oval; ciliation uniform. This small, reddish-pigmented freshwater species is well defined due to the redescriptions by Foissner (1980) and Song and Wilbert (1989). Foissner et al. (1992) reviewed the data available on its morphology and ecology. Stentor igneus lacks symbiotic algae and has two synonyms: S. roseus and S. ruber. Measurements. 200-400 um long.
Photos / Sounds
What
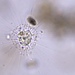
Genus TrithigmostomaObserver
juanloredoDescription
Water sample collected from the river San Pedro, Congregación Ortíz, Rosales, Chihuahua, at the indicated location.
The sample was collected on January 5th at 15:42. The images were acquired on January 7th.
Photos / Sounds
Observer
amayakanDescription
Video (x5 speed): https://youtu.be/0qTAE73Mgt4
Photos are snapshots of the same individual in the video
Size: see scale bar
Site of collection: Pavilion, Takamori Higashi Park (a freshwater habitat), Sendai, Japan
Date of collection: Aug. 8th 2021
Weather: Rainy
Water temp.: 29.0°C
pH 6.6
Date of observation: September 12th 2021 (the collected sample in a plastic container was left near a window out of direct sunlight at room temperature until observation).
Bright field observation using a Wraymer microscope (model BX-3500TL, Osaka, Japan) equipped with a Floyd-2 HDMI ethernet digital camera (Wraymer, Osaka, Japan). The accuracy of the scale bar was confirmed by using a stage micrometer glass slide (1 div. = 10 µm; Wraymer, Osaka, Japan) at each magnification.
Photos / Sounds
What
Genus ZosterodasysObserver
amayakanDescription
Video: https://youtu.be/NVS2DjYjfX8
Photos are snapshots of the same individual in the video.
The cell measures 77 µm x 30 µm
Site of collection: Katsurashima Ryokuchi south pond (a freshwater habitat), Sendai, Japan
Date of collection: February 11th 2022
Weather: Sunny
Water temp.: 5.2°C
pH 6.6
Date of observation: February 12th 2022 (the collected sample in a plastic container was left near a window out of direct sunlight at room temperature until observation)
Bright field observation using a Wraymer microscope (model BX-3500TL, Osaka, Japan) equipped with a Floyd-2 HDMI ethernet digital camera (Wraymer, Osaka, Japan).
Photos / Sounds
Observer
amayakanDescription
video (x1 speed): https://youtu.be/wCv0HsU80Bo
Photos are snapshots of the same individual in the video
Cell size: 50 µm x 43 µm
Site of collection: Katsurashima Ryokuchi south pond (a freshwater habitat), Sendai, Japan
Date of sample collection: September 18th 2021
Weather: Rainy
Water temp.: 21.3°C
pH 6.7
Date of observation: September 23rd 2021 (the collected sample in a plastic container was left near a window out of direct sunlight at room temperature until observation)
Bright field observation using a Wraymer microscope (model BX-3500TL, Osaka, Japan) equipped with a Floyd-2 HDMI ethernet digital camera (Wraymer, Osaka, Japan). The accuracy of the scale bar, which was set according to the guidance of the manual of the software (Spectman) provided for the camera, was confirmed by using a stage micrometer glass slide (1 div. = 10 µm; Wraymer, Osaka, Japan) at each magnification.
What
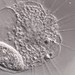
Genus NebelaObserver
nickbelliveauDescription
From mosses and liverworts in damp roadside ditch. 400x magnification.
Stats
- 7511